A HIP REPLACEMENT LAWSUIT CAN HELP YOU AND YOUR LOVED ONES RECOVER
If your health or that of a loved one has been compromised by the failure of OMNI K2™ Hip replacement, you may be entitled to compensation. You can pursue a hip replacement lawsuit with our Oklahoma City, OK, attorneys, who can help you hold the manufacturer responsible. At Tawwater Law Firm, our lawyers can use their extensive experience in product liability and defective medical devices to aggressively pursue just compensation for injuries you have suffered because of compromised hip replacements. If you or a loved one has suffered injuries as a result of these hip implants, do not hesitate to contact our law firm today to schedule a consultation.
About OMNI K2™ Hip Implants
The OMNI K2™ Hip System, often called the Apex K2 hip implant, is an adjustable hip replacement system that was approved by the FDA in 2004. These implants were approved for use in patients with rheumatoid arthritis, congenital deformities, osteoarthritis, and fractures. Some doctors also used the OMNI K2™ to replace other faulty implants during revision surgeries.
This hip replacement system was approved through the FDA’s controversial 510(k) application process. This process states that testing is not required for devices that are comparable to another approved device that is already on the market. This loophole allowed OMNI to completely circumvent any safety testing for the device. In addition to the lack of safety testing, the device itself was poorly designed. Although adjustable implants provide patients with a customized fit, they are more susceptible to breakage and dislocation. The patient may experience severe pain and a fractured bone as a result. The poor design and lack of testing has also resulted in further complications for many victims and additional health problems.
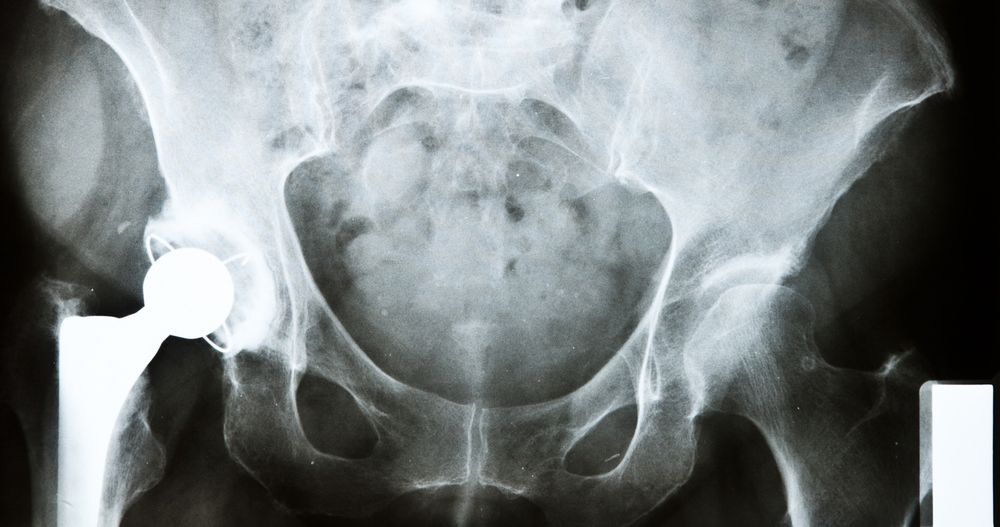
Complications from Defective Hip Implants
Recent studies have shown the OMNI K2™ hip replacement system has a higher-than-average failure rate and higher-than-average revision surgery rates. Complications from these implants may include:
- Metallosis: A type of metal poisoning caused by the metal parts of the implant rubbing together. Symptoms may include hearing impairment, vision impairment, cognitive impairment, heart failure, infection, and more. Some of these symptoms may dissipate following revision surgery, but hearing or vision loss could be permanent.
- Breakage or dislocation of the implant: This breakage can cause a fracture of the femur and severe discomfort, which requires emergency revision surgery, and can weaken your remaining bone tissue. Dislocation can also lead to severe discomfort and may require further surgery to correct.
- Negative reaction in the surrounding tissues: Irritation and rejection of the material can lead to non-cancerous lesions and discharge.
Defective Medical Devices Lawsuits
Due to the extremely high rate of failure and revision surgeries, Australia pulled the OMNI K2™ implants from the market in 2012. Unfortunately, no such warnings have been issued in the United States, and there has been no official recall of the device. Often, the best option for those who have suffered because of these devices is to pursue a defective medical device lawsuit, which is a type of product liability case. These lawsuits hold the manufacture accountable for their negligence.
As your legal team, Tawwater Law Firm can seek full compensation for any medical expenses associated with your revision surgery or any other complications.
In this case, the manufacturer, OMNI, can be held liable for the complications associated with their poor design and failure to adequately test the device. As your legal team, Tawwater Law Firm can seek full compensation for any medical expenses associated with your revision surgery or any other complications. We can also seek compensation for lost wages, pain and suffering, and a reduced quality of life.
Seek Experienced, Proven Representation
If you or a loved one has suffered complications as a result of defective hip implants, contact Tawwater Law Firm today to schedule a free consultation. We will review your case and explain the legal options available to you.